Researchers at Rice and Politecnico di Milano in Italy have developed a better understanding of how light interacting with nanostructures can drive chemical reactions and save energy in the process.
“The chemical industry is typically fed by fossil fuels and represents one of the largest energy consumers in the world. By shining light on metal nanoparticles, we are able to control these chemical reactions, leading to improved energy efficiency and reduced cost,” said Alessandro Alabastri, assistant professor of electrical and computer engineering (ECE) at Rice and corresponding author of the research.
Alabastri and his colleagues have published their findings, “Quantifying Ultrafast Energy Transfer from Plasmonic Hot Carriers for Pulsed Photocatalysis on Nanostructures,” in ACS Nano, a publication of the American Chemical Society.
Researchers have known that light can stimulate chemical reactions – a process known as photocatalysis – but assessing the efficiency of the process and understanding the mechanisms involved remain a challenge.
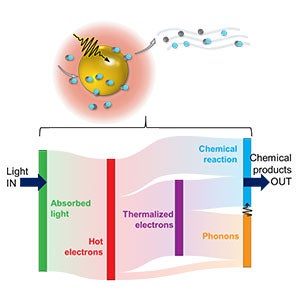
In their latest work, Alabastri and his co-authors have developed a modeling approach to predict the rate at which photocatalytic reactions occur on extended nanostructures, isolating the role of highly energetic “hot electrons” from thermalized electrons and phonons.
“Despite the promising advances, a thorough description of the ultrafast dynamics of excited carriers, including hot electrons, and the resulting thermal effects across extended plasmonic nanostructures in the presence of adsorbed molecules has remained elusive,” said Andrea Schirato, Marie Curie Fellow at Politecnico di Milano, visiting research scholar at Rice and first author of the article.
“Our simulations can be adapted to a large variety of nanoparticle-molecule electron transfer models and serve as tools to design, engineer and optimize nanophotonics-based energy conversion systems,” said Stephen Sanders, Rice Academy Fellow, Research Fellow at the Army Research Laboratory and co-first author.
Their co-authors are Remo Proietti Zaccaria, Italian Institute of Technology; Peter Nordlander, Wiess Chair and professor of physics and astronomy, ECE and of materials science and nanoengineering; Giuseppe Della Valle, Politecnico di Milano.
The work is funded in part by a Marie Skłodowska-Curie Action grant from the European Union’s Horizon Europe research and innovation program. Alabastri is a member of the Smalley-Curl Institute, the Rice Advanced Materials Institute and the Laboratory for Nanophotonics.

